Ocean Acidification Adalah. It is the long-term change in seawater chemistry due to the absorption of carbon dioxide from the atmosphere. Scientists have observed that the ocean is becoming more acidic as its water absorbs carbon dioxide from the atmosphere.

Ocean acidification is a people problem, too. Thus, ocean acidification not only reduces surface ocean pH, but also reduces the availability of carbonate ion. Ocean acidification is a complex issue that is being studied and addressed from several viewpoints, in various locations, and for different issues.
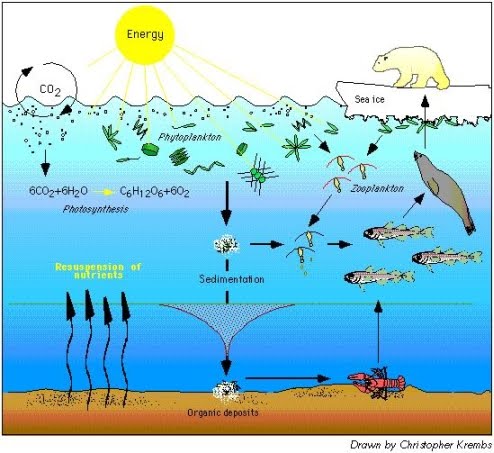
Many factors contribute to rising carbon dioxide levels. Many of these farmers and fishermen have operated their coastal businesses for decades, with new family members taking. When carbon dioxide enters the ocean, it combines with seawater to produce carbonic acid, which increases the acidity of the water, lowering its pH.
These chemical reactions are termed "ocean acidification" or "OA" for short. Changes in ocean chemistry can affect the behavior of non-calcifying organisms as well. Ocean acidification is a global threat to the world's oceans, estuaries, and waterways.
The present-day pH of seawater is highly variable, and a single organism can cope with. The pH refers to the potential or power of hydrogen. The ability of some fish, like clownfish, to detect predators is decreased in more acidic waters.
Many of these farmers and fishermen have operated their coastal businesses for decades, with new family members taking. Location of planned OA monitoring and research sites and affiliated NOAA labs. Ocean acidification is a people problem, too.
Ocean acidification affects various Earth system processes and phenomena, including: Changing biomass and productivity, which can alter species interactions. Calcium carbonate minerals are the building blocks for the skeletons and shells of many marine. In this presentation, participants will dive deep into Data in the Classroom's Ocean Acidification Module to explore the processes that cause acidification, examine data from across the globe and take a virtual tour of the new web-based curricular modules and data tools.
Currently, the burning of fossil fuels such as coal, oil and gas for human industry is one of the major causes. Ocean acidification is mainly caused by carbon dioxide gas in the atmosphere dissolving into the ocean. Ocean acidification has become a threat just in the two hundred years or so since the advent of the Industrial Revolution, and there is strong evidence it is a direct result of human activity.
Calcium carbonate minerals are the building blocks for the skeletons and shells of many marine. As the amount of carbon dioxide in the atmosphere increases, the amount of carbon dioxide absorbed by the ocean. These chemical reactions are termed "ocean acidification" or "OA" for short.
Location of planned OA monitoring and research sites and affiliated NOAA labs. Ocean acidification itself is a fairly simple chemical process. Ocean acidification is mainly caused by carbon dioxide gas in the atmosphere dissolving into the ocean.
Ocean acidification impacts on fish and seaweeds. The Short Answer: Ocean acidification is a change in the properties of ocean water that can be harmful for plants and animals. Many of these farmers and fishermen have operated their coastal businesses for decades, with new family members taking.
Ocean acidification is a complex issue that is being studied and addressed from several viewpoints, in various locations, and for different issues. This makes it more difficult for marine organisms, such as coral and some plankton, to form their shells and skeletons, and existing shells may begin to dissolve. Ocean acidification is sometimes called "climate change's equally evil twin," and for good reason: it's a significant and harmful consequence of excess carbon dioxide in the atmosphere that we don't see or feel because its effects are happening underwater.
Ocean acidification is mainly caused by carbon dioxide gas in the atmosphere dissolving into the ocean. This leads to a lowering of the water's pH, making the ocean more acidic. The pH refers to the potential or power of hydrogen.
This makes it more difficult for marine organisms, such as coral and some plankton, to form their shells and skeletons, and existing shells may begin to dissolve. The ability of some fish, like clownfish, to detect predators is decreased in more acidic waters. When carbon dioxide enters the ocean, it combines with seawater to produce carbonic acid, which increases the acidity of the water, lowering its pH.
Location of planned OA monitoring and research sites and affiliated NOAA labs. It is often called "climate change's evil twin" and is projected to grow as carbon dioxide continues to be emitted into the atmosphere at record-high levels. This leads to a lowering of the water's pH, making the ocean more acidic.
From the Atlantic to the Pacific and all around the world, people who work on the water are facing the impacts of acidification. Ocean acidification is a complex issue that is being studied and addressed from several viewpoints, in various locations, and for different issues. Many of these farmers and fishermen have operated their coastal businesses for decades, with new family members taking.
With increased use of fossil fuels, that number is now.
Calcium carbonate minerals are the building blocks for the skeletons and shells of many marine.
Ocean acidification reduces the amount of carbonate, a key building block in seawater. Ocean acidification impacts on fish and seaweeds. Fundamental changes in seawater chemistry are occurring throughout the world's oceans.